The GENEROUS Model and the Evolution of Most Favored Nation Pricing Strategies in the Medicaid Prescription Drug Market
The federal government and state Medicaid programs are currently navigating a significant shift in pharmaceutical procurement through the implementation of the Generating Cost Reductions for U.S. Medicaid (GENEROUS) Model. Launched by the Centers for Medicare & Medicaid Services (CMS) Innovation Center in January 2026, this initiative represents the operationalization of "Most-Favored-Nation" (MFN) drug pricing. The model is built on the fundamental premise that the United States, as a major global purchaser of pharmaceuticals, should not pay significantly higher prices for prescription drugs than other comparable high-income nations. While spending on prescription drugs constitutes a relatively modest portion of total Medicaid expenditures, the rapid growth in this sector has prompted a strategic pivot toward international benchmarking as a primary tool for cost containment.
Under the GENEROUS framework, CMS seeks to align domestic Medicaid net prices with the prices paid in eight peer countries: the United Kingdom, France, Germany, Italy, Canada, Japan, Denmark, and Switzerland. This shift comes at a time when research consistently indicates that U.S. drug prices are approximately three times higher than those in other OECD nations, with brand-name drug prices reaching as high as 4.22 times the international average. By negotiating supplemental drug rebates based on these international benchmarks, the administration aims to secure substantial savings for both federal and state taxpayers.
A Chronology of Implementation and Key Milestones
The path toward the GENEROUS model was paved by a series of executive initiatives and bilateral agreements between the Trump administration and major pharmaceutical manufacturers. The first of these landmark deals was announced in September 2025 with Pfizer, followed by a second agreement with AstraZeneca in October 2025. By December 2025, the administration had secured the largest set of MFN commitments to date, involving a total of 17 pharmaceutical companies, including Regeneron.
The official launch of the GENEROUS model in January 2026 initiated a structured application window for both manufacturers and state Medicaid agencies. While originally set with tighter deadlines, CMS has extended the application period twice to accommodate the complexity of the transition. Manufacturers now have until June 11, 2026, to apply for participation. States, which participate on a voluntary basis, have until July 31, 2026, to submit their applications, with a final deadline of August 31, 2026, to execute formal participation agreements.
The model is designed as a five-year demonstration project running through 2030. During this period, CMS will monitor the impact on health care spending and patient access. The voluntary nature of the model allows for flexibility; however, the administration has utilized external economic levers—specifically the threat of pharmaceutical tariffs—to encourage manufacturer compliance. Companies that signed MFN agreements were granted a three-year reprieve from these tariffs, a move that bolstered initial participation but leaves long-term manufacturer engagement subject to future negotiations.
The Technical Framework: Calculating the MFN Price
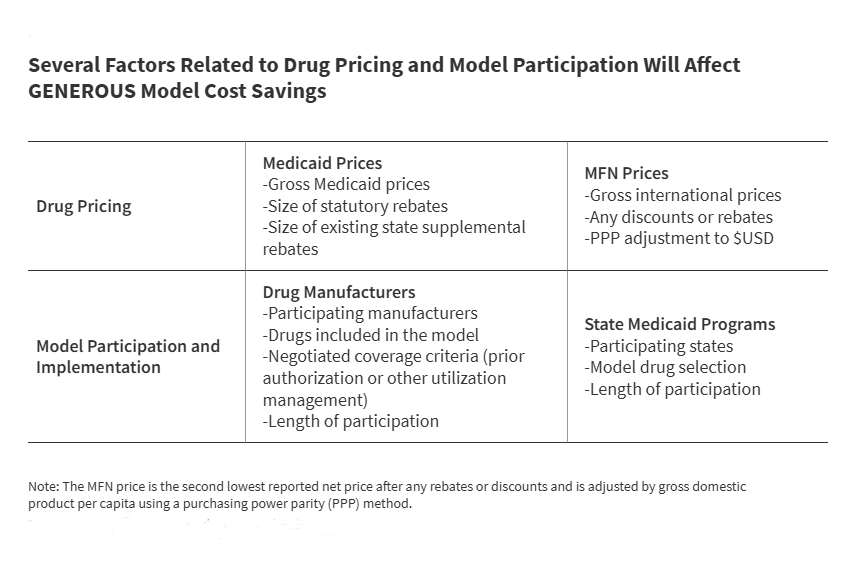
The GENEROUS model utilizes a sophisticated methodology to determine the "MFN price," which serves as the benchmark for Medicaid supplemental rebates. This process begins with manufacturers providing international pricing data for "model drugs," which include single-source or innovator multiple-source brand-name medications.
CMS identifies the MFN price by locating the second lowest reported net price among the eight reference countries. To ensure an equitable comparison, this price is adjusted for inflation and economic differences using a purchasing power parity (PPP) method and gross domestic product (GDP) per capita metrics. Once the MFN price is established, CMS calculates the necessary supplemental rebate required to bring the Medicaid net price into alignment with this international benchmark.
Crucially, the model introduces "uniform coverage criteria." In exchange for the lower MFN-based pricing, states must adopt standardized utilization controls, such as specific prior authorization requirements or step therapy protocols. This standardization is intended to reduce the administrative burden associated with negotiating hundreds of individual state-level supplemental rebate agreements (SRAs). However, it also requires states to cede some control over their preferred drug lists (PDLs), a trade-off that is currently being analyzed by state pharmacy directors nationwide.
Financial Projections and Economic Impact Analysis
A recently released White House report provides an optimistic outlook for the fiscal impact of the GENEROUS model. The report estimates that a voluntary MFN framework within Medicaid could generate $64.3 billion in savings over a 10-year horizon. This equates to an average annual saving of $6.43 billion, representing approximately 14% of current annual Medicaid prescription drug spending.

However, economists note that the savings trajectory is unlikely to be linear. Initial savings are expected to be substantial as U.S. prices are forced down to meet international levels. Over time, these savings may diminish due to several market factors. One primary concern is "price creep" in the reference countries; as international markets become benchmarks for the massive U.S. Medicaid program, manufacturers may seek to increase prices in Europe or Canada to protect their U.S. revenue margins.
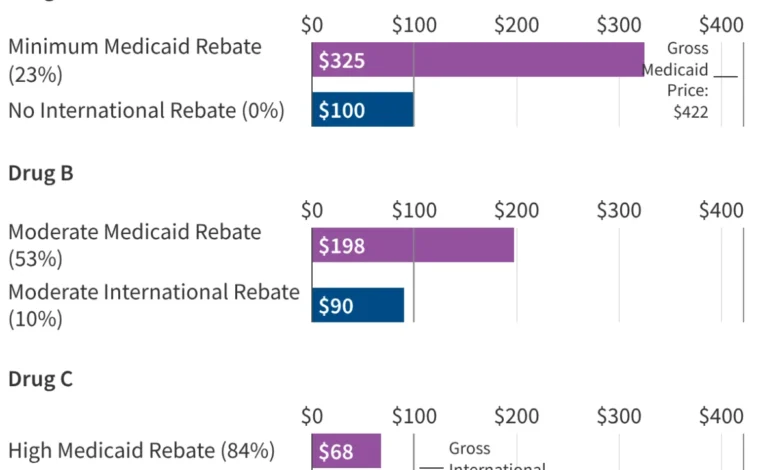
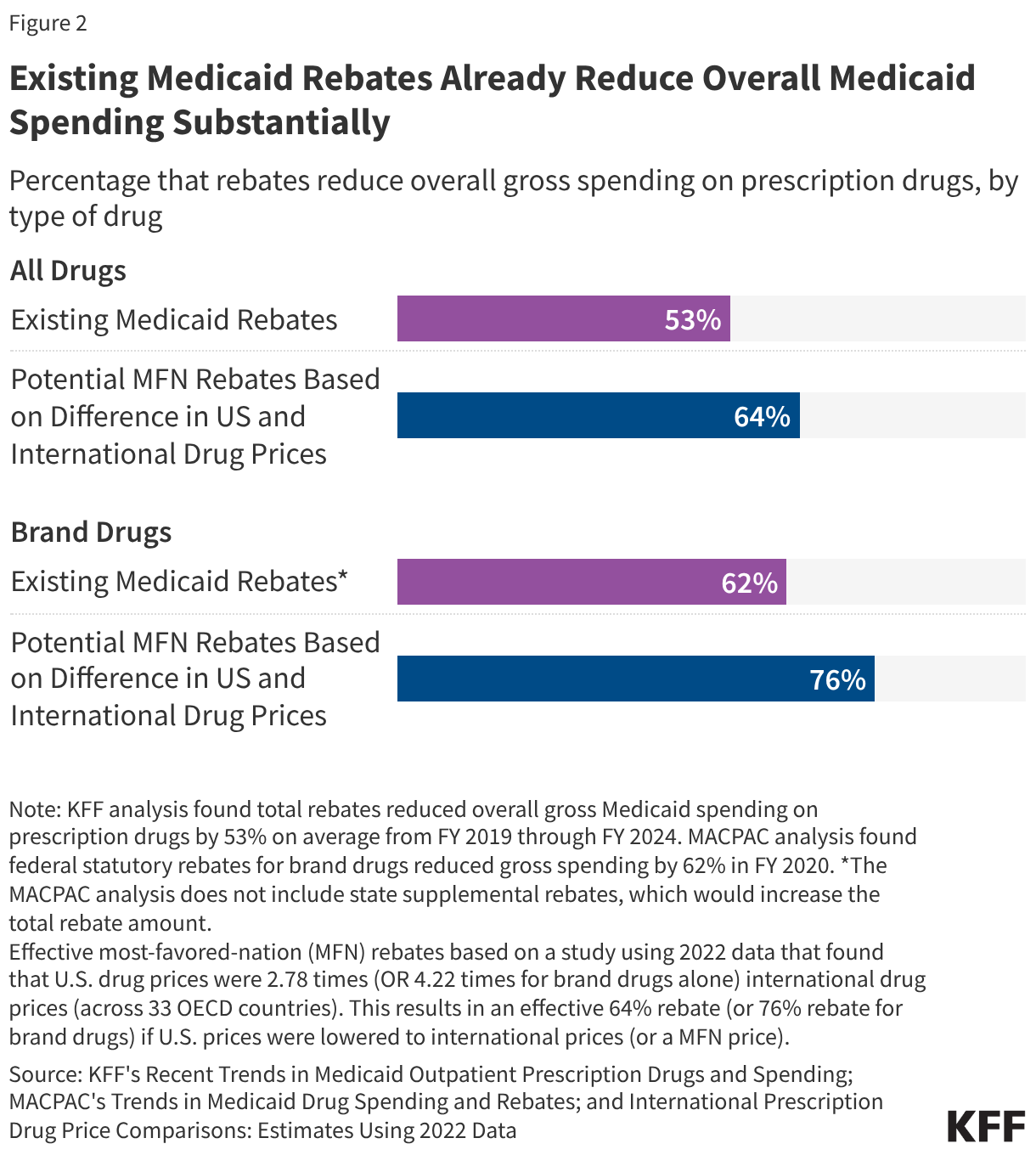
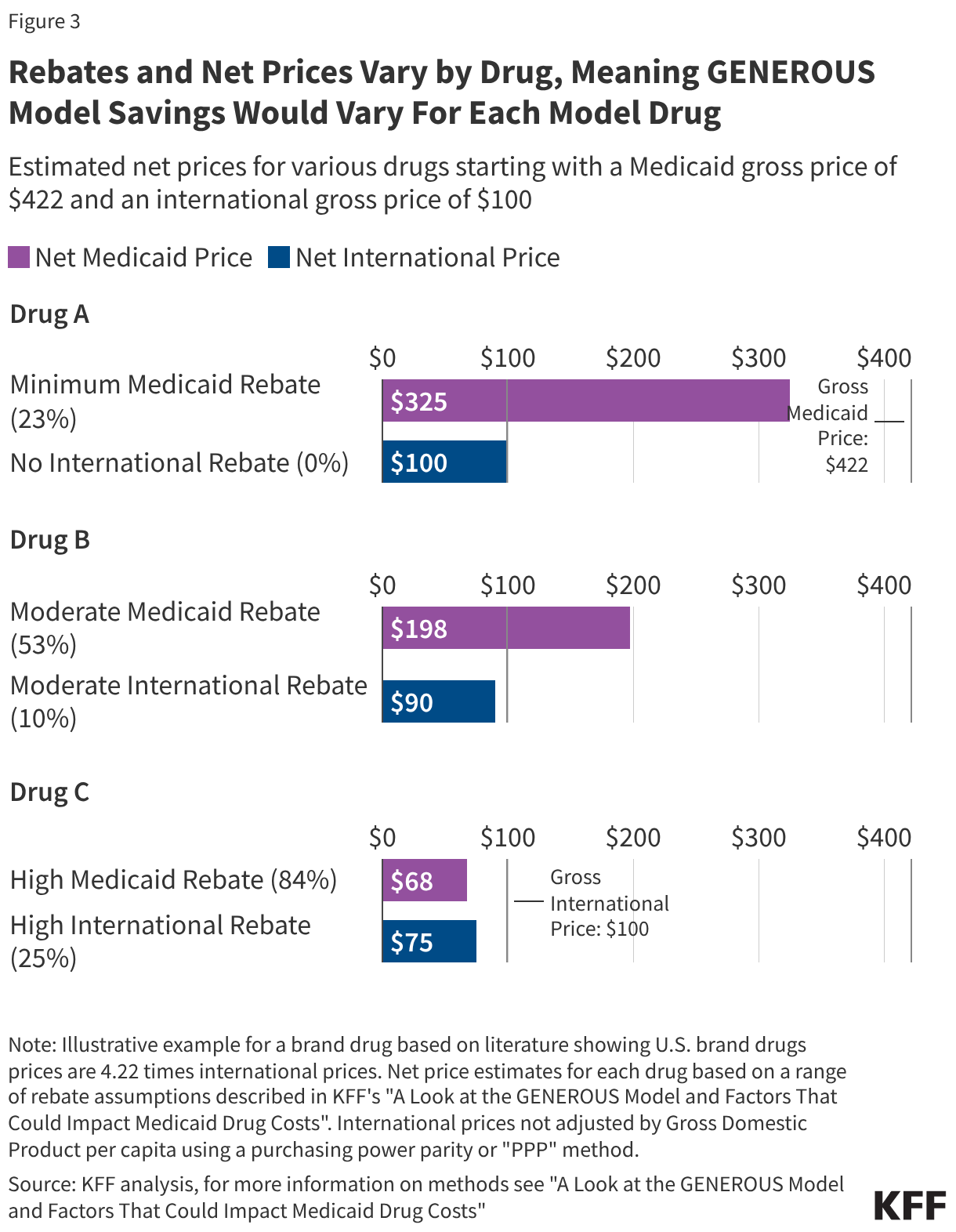
Furthermore, the impact of the GENEROUS model must be viewed through the lens of the existing Medicaid Drug Rebate Program (MDRP). Under current law, manufacturers already provide significant rebates to the Medicaid program, consisting of a basic rebate and an inflationary component. Between FY 2019 and FY 2024, these rebates reduced gross Medicaid drug spending by an average of 53%. For brand-name drugs, the total rebate can be even higher, with some estimates suggesting a 62% to 77% reduction from the list price. Because Medicaid already receives the "best price" available to most commercial payers, the incremental savings provided by the GENEROUS model will depend entirely on how much lower the international MFN price is compared to the already-discounted Medicaid net price.
Variability Across States and Therapeutic Classes
The impact of the GENEROUS model will not be uniform across the United States. Medicaid programs are administered at the state level, and the effectiveness of current cost-containment strategies varies widely. Data from the Medicaid and CHIP Payment and Access Commission (MACPAC) for FY 2024 reveals that while the national average rebate is 53%, some states like Delaware and Wyoming see rebates exceeding 90% of gross spending. Conversely, states like Oregon and Virginia see rebates of less than 40%.
States with already high rebate levels may find that the GENEROUS model offers little additional financial benefit, while states with lower current rebates could see a windfall. Additionally, the concentration of spending on a few high-cost drugs complicates the savings outlook. KFF analysis shows that just five drugs—Biktarvy, Humira, Stelara, Dupixent, and Ozempic—account for 10% of all Medicaid drug spending. The top 50 drugs account for more than one-third of total spending.
The success of the GENEROUS model hinges on whether these "blockbuster" drugs are included in the MFN negotiations. For example, a newer drug like Biktarvy might have a relatively low existing Medicaid rebate (estimated at 24% in its early years), making it a prime candidate for significant MFN savings. In contrast, an older drug like Eliquis may already be subject to such high inflationary rebates that the Medicaid program pays nearly nothing for it, rendering the MFN benchmark redundant.
Stakeholder Reactions and Legislative Oversight
The implementation of the GENEROUS model has met with a mix of cautious optimism from state budget offices and intense scrutiny from legislative bodies and the pharmaceutical industry. Senate Democrats, led by Finance Committee Chairman Ron Wyden, have recently pressed pharmaceutical companies for greater transparency regarding the "secret deals" negotiated with the administration. In formal inquiries, lawmakers have requested evidence that these MFN agreements will result in genuine cost savings for taxpayers rather than merely serving as a mechanism for companies to avoid tariffs.
Manufacturer participation remains a point of contention. While 17 major firms have joined, the voluntary nature of the model means that many drugs remain outside the MFN framework. The administration’s recent imposition of tariffs on patented pharmaceutical products for non-participating companies represents an aggressive use of executive power to compel participation, a move that trade experts suggest could be challenged in international trade forums or domestic courts.
Patient advocacy groups have also raised concerns regarding the "uniform coverage criteria." While lower prices are welcomed, there is apprehension that standardized prior authorization or step therapy could create new barriers to access for Medicaid enrollees with complex chronic conditions. CMS has countered these concerns by stating that the model includes rigorous monitoring and evaluation to ensure that health outcomes are not compromised in the pursuit of fiscal savings.
Broader Implications for the Global Drug Market
The GENEROUS model represents more than just a domestic policy shift; it is a significant intervention in the global pharmaceutical economy. By formally linking U.S. Medicaid prices to international benchmarks, the U.S. is leveraging its market power to challenge the long-standing status quo where American patients and taxpayers subsidize global research and development through higher prices.
As the model progresses toward its 2030 conclusion, its influence may extend beyond Medicaid. If successful, the MFN approach could serve as a blueprint for Medicare or private insurance markets. However, the potential for unintended consequences is high. If manufacturers respond by delaying drug launches in the eight reference countries or by raising prices globally, the GENEROUS model could trigger a restructuring of international drug pricing agreements.
For now, the focus remains on the August 2026 deadline for state participation. The number of states that choose to opt in will be the first true metric of the model’s perceived value. As state legislatures face mounting budget pressures and potential federal Medicaid cuts, the promise of $64 billion in savings may prove to be an irresistible incentive, potentially making the GENEROUS model the most significant change to Medicaid pharmacy policy in a generation.